
Rutherford model, also called Rutherford atomic model, nuclear atom, or planetary model of the atom, description of the structure of atoms proposed (1911) by the New Zealand-born physicist Ernest Rutherford. Based on these results, Rutherford proposed the nuclear model of the atom. Alpha particles are deflected at tiny concentrated center while for most of the part these rays go undeflected shows having large. Rutherford's gold foil experiment showed that the atom is mostly empty space with a tiny, dense, positively-charged nucleus. The Geiger-Marsden experiment (also called the Gold foil experiment or the Rutherford experiment) was an experiment done by Hans Geiger and Ernest Marsden in 1909, under the direction of Ernest Rutherford at the Physical Laboratories of the University of Manchester which led to the downfall of the plum pudding model of the atom. Rutherford model of atom contained the features of a relatively high central charge concentrated into very small volume in comparison to the rest of the atom and this central region was named nucleus of the atom. What was Rutherford experiment and what did he discover? In 1911, he was the first to discover that atoms have a small charged nucleus surrounded by largely empty space, and are circled by tiny electrons, which became known as the Rutherford model (or planetary model) of the atom. When did Ernest Rutherford contribute to the atomic theory? Now, we have got the complete detailed explanation and answer for everyone, who is interested!  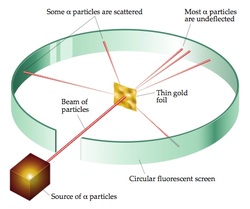 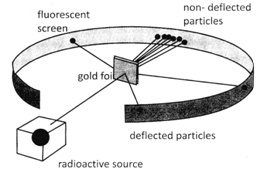
This is a question our experts keep getting from time to time. Who is ernest rutherford and what did he do?
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
